Heal Italia represents the first Italian Foundation that coordinates a qualified multidisciplinary network of Universities, IRCCS and Companies, scientists, technologists and young researchers who share knowledge, research and innovative technologies in order to bring our National Health System into the contemporary era of Medicine of Precision through new methods, new services and above all an important network of clinical data to support translational research for advanced diagnoses and therapies in the fight against cancer and cardiovascular, metabolic and rare diseases. The overall cost amounts to €118,765,450.00 and the MUR subsidy amounts to €114,700,000.00. The cascading tenders provide for 10% of the project costs.
The HEAL ITALIA Project aims to strengthen the research and innovation chain in the field of Precision Medicine, also through active participation in the development of value chains considered strategic for the country.
The Project, admitted to financing with concession decree prot. 1559 of 11 October 2022, with a financial endowment of 114.7 million, represents the first Italian multidisciplinary network of universities, IRCCS and companies, in which scientists, technologists, clinicians and young researchers share knowledge, research and innovative technologies in order to to bring the Research System into the contemporary era of Precision Medicine.
Personalized and Precision Medicine represent a shift from a one-size-fits-all approach to an optimized strategy for the prevention, diagnosis and treatment of diseases for each person, based on their unique characteristics. Consequently, this approach places the patient at the center of the healthcare system, aiming for optimal management of the disease and/or predisposition to the disease. The benefits of Precision Medicine are proven by recent developments in areas such as: diagnostic tests, Omic technologies, analysis of molecular mechanisms, real-time monitoring of disease-associated parameters and compliance with prescribed medicines.
In this context, the HEAL ITALIA Project is conceived with a holistic interdisciplinary vision, capable of combining fundamental and translational research with technology transfer, exploiting the capabilities of the main players in the academic, clinical and private sectors. The Project envisages that the research & development activities will be carried out at eight Spokes, coordinated by the HUB, which acts as the implementing body.
The Hub is in fact the entity responsible for the initiation, implementation, management and coordination of the Extended Partnership. It represents the sole point of contact for the MUR, receives the tranches of concessions granted, verifies and transmits to the MUR the reporting of the activities carried out by the Spokes and their affiliates.
The Spokes represent the nursery of the project, i.e. the place where the activities are carried out. HEAL ITALIA is organized into eight spokes coordinated by eight Public Universities (Tor-Vergata University, Alma Mater Studiorum Bologna, University of Palermo, Sapienza University of Rome, University of Milan Bicocca, University of Modena and Reggio Emilia , Polytechnic University of Marche and University of Pisa) which as Spoke make use of the collaboration of affiliated entities for the implementation of research activities within their competence. The eight Spokes represent eight interregional networks that cover all areas of interest of the Research Program.
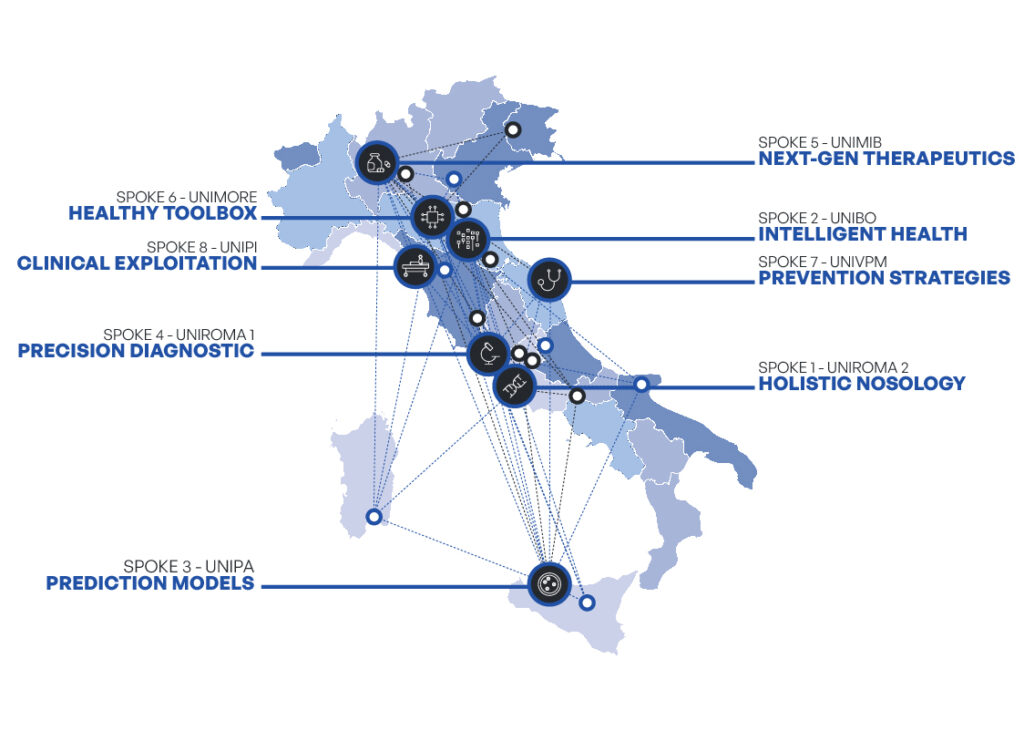
The objective of the HEAL ITALIA research project is the creation of research chains, in which public and private sectors operate synergistically in order to support the implementation of processes and the creation of preparatory products for Precision Medicine.
The overall objective of the project is therefore to provide new, cost-effective and evidence-based predictive and non-invasive diagnostic pathways for faster, earlier, more precise, accessible and economical prediction, detection and monitoring of monogenic (rare) diseases. ), polygenic (cardiovascular and metabolic) and cancer, as well as to identify innovative and effective therapeutic approaches. The HEAL ITALIA project also aims to identify and reduce inequalities (extremely large between Northern and Southern Italy) by developing an interregional collaboration roadmap to define evidence-based pathways that can be easily used in clinical practice. The Mission of the HEAL ITALIA Research Program is therefore to facilitate the transition towards efficient methods and tools in terms of costs and time, which allow early diagnosis and frequent screening of patients, two of the cornerstones of approaches to precision medicine . The activities are therefore aimed at acquiring new knowledge and tools that are also useful for the development of new processes and new products. In this context, a standardized approach to evaluating technologies intended for the healthcare world will guide the introduction of these technologies into clinical practice.

